We are familiar with the concept of our grass plants being composed mostly of water (75-85%), but what else is in a grass plant? The answer is that the dry matter of the plant is made up of a mix of 16 elements, commonly referred to as the essential nutrients. We describe them as essential because the plant can't exist or complete its life cycle if any of these nutrients are lacking to any great degree.
Tag: soil chemistry
Can we really trust the fertiliser trade?
The somewhat uncomfortable truth is that we as greenkeepers are actually part of the massive worldwide agriculture industry.
Very few products or technologies ever see the light of day based purely on the needs of bowling clubs or even the much larger golf segment of the fine turf and sports industry.
No, most of the things we use are direct descendants of agricultural or other industrial products or at least are supported by agriculture’s huge global enterprise.
Every chemical pesticide we use is a direct copy of a product which has a use in growing crops; every fertiliser product is a result of agricultural research and manufacturing processes; even our mowers are based on a machine originally used for trimming in the massive Victorian carpet and textile industry.
Its very interesting to see as part of this discussion the current and on-going dilemma that faces farming. The burgeoning world population means that we have to grow more and more food on fewer and fewer hectares of ground. This ensures that agriculture will continue to be a cutting edge area of scientific research and that as a result we can look forward to continued and constant enticement to try out a multitude of new products and techniques on our bowls greens in the future.
However, there is one train of thought in agriculture and in society in general that is much closer to the one we need to nurture for the assured excellence of our bowls greens in the future, and that is ecological sustainability.
To accompany this goal of sustainability within agriculture there is a renewed interest in the common sense concept of healthy living soil and that is where we need to start on our road to an excellent,
high performance bowling green.
Incidentally, there is an excellent program on the BBC iplayer that explains the mechanics of soil ecology very well. You can still access it here.
Sub-Surface Requirements for a High Performance Bowls Green
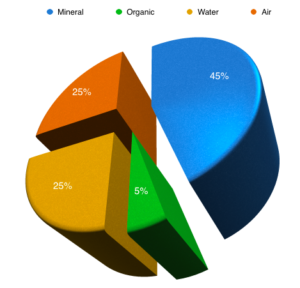
The Sub-Surface Requirements for a High Performance Bowls Green are pretty much set in stone. Get these right and you’re well on your way to a Performance Bowling Green.
First of all then, have a look at the diagram above. This represents the ideal make up of a performance bowls green’s rootzone.
Do you see anything remarkable?
Well, when I explain this to my clients for the first time, many of them are surprised to say the least.
If you look closely at the diagram you will see that the ideal green will be 50% nothing; yes space, just air cavities within the soil. Now, of course I am not going to tell you to get rid of half your green to achieve this, but next week I am going to share with you a program whereby you can get close to this ideal situation of ½ solids and ½ space in your green.
Of course the space isn’t just nothing; half of the space consists of “micro-pores” and half is “macro-pores”. Put simply the micro-pores contain water and the macro-pores contain air. This is very important to understand and is one of the least understood concepts within sportsturf maintenance.
The “nothing” element of the ideal green is the most important factor to get right, because this is where we get the balance between speedy drainage and good growing conditions and it is due to a fundamental misunderstanding of this concept that a very large number of UK bowling greens are in poor condition and can’t be prepared for performance consistently. The only saving grace for these greens is that the UK summer is also very inconsistent and sometimes acts in their favour; so that we occasionally get a very good season’s bowling when the “green has never been better”. This is a false reading in most cases and the problem is exacerbated by the club attributing this success to the latest fad program.
The green which has a well balanced soil as described and illustrated above will naturally:
- sustain a firm, fast surface with a minimal input maintenance program
- sustain a healthy sward of fine grasses
- sustain a high, year-round population of soil microbes
- provide a natural cycle of nutrient release from soil organisms and micro-organisms (microbes) working on fresh organic matter (thatch).
- resist compaction and therefore resist:
- shallow rooting
- annual meadow grass ingress
- flooding and puddling
- head skinning
- retain the optimum amount of soil water for healthy growth with minimum requirement for artificial irrigation.
- drain reasonably quickly after excessive rainfall.
- retain the optimum amount of plant available nutrition
- sustain a soil pH within the optimum range for fine turf
- resist attack from fungal diseases
- resist the onset of Localised Dry Patch and other soil and turf disorders
- maintain a tight, dense sward with an upright growth habit which will reduce ingress of moss, weeds and weed grasses.
- resist localised settlement and bumpiness due to excessive thatch and erratic thatch decomposition
Comprehensive action plan for achieving the above included in the book below. It costs less than a bag of cheap fertiliser.
Transforming your Bowls Green-the knowledge.
I keep going on about healthy living soil and healthy living turf and healthy living bowling greens on this site.
In my eBook Performance Bowling Greens, a practical guide, I focus heavily on the process of turning bowling greens back in to healthy living eco systems that are pretty much self sustaining.
By self sustaining I mean that they are in a condition that allows them to be set up and prepared for play in a consistently high performance manner with no pesticides and minimal on-going in-put costs.
This relies on the green being converted from a barren, sand laden, resource swallowing beast that is unpredictable at best and disappointing and expensive at worst.
This conversion process demands two qualities in those who seek to deliver a Performance Bowling Green:
Knowledge, that you are doing the right thing and the Patience to spend the time following the program.
Performance Bowling Greens spells out this process in great detail, but here in basic terms are the key points of knowledge that are important to a successful transition:
- Soil micro-life is critically important; this refers to the microscopic life in all soils and includes beneficial fungi which help plants to assimilate nutrients (which fungicides get rid of), and soil microbes, which help to turn organic matter (mainly thatch) into plant useable nutrient ions.
- Every time you add sand to the green, the abundance of soil micro-life reduces; sand is inert.
- Sandy soils generally have a lower Cation Exchange Capacity (the ability to retain the plant useable nutrient ions created by the soil microbes).
- There are two distinctly different kinds of pore space in soils; micro or capillary pore space where plant roots take up moisture and nutrients; and macro or aeration pore space where drainage occurs (to stop the soil from being saturated with water), and oxygen (critical to maintaining a large population of soil microbes) is held.
- Compaction ruins the structure of the soil and reduces aeration pore space, oxygen content and soil microbe populations. This is why compacted greens seem to need more and more fertiliser. Compaction + More Fertiliser = Thatch and so the downward spiral into the Circle of Decline begins.
These are 5 guiding principles if you like for a consistently high performance green. You will notice that this is not a list of things to do, but a list of key things to keep in mind, to focus on as we patiently go about the transition process. You can be confident that if you are heading in this direction you are heading in the right direction regardless of how long the road is.
And you can be confident that the view will improve (in the shape of performance) around every corner of that road.
Bio Liquid Fertilisers- their role in Performance Bowls Greens
I often mention that for a Performance Bowling Green, you should be using Bio Liquid Fertilisers and in an earlier article I briefly discussed some of the key characteristics exhibited by what I would term a Performance Green and these were:
- A green that performs well from the get go every year.
- A green that is consistent throughout the season
- A green that is economical to maintain
- A green that fights off disease, drought and cold largely on its own.
- A green that your members will relish playing on
I then went on to give a couple of hints for starting the transition from a conventionally maintained green to a Performance Green. These were to stop routinely applying sand based top-dressing and to start as soon as possible to use Bio-Liquid fertilisers throughout the growing season.
The top-dressing debate has raged on this site for a long time now and I think I’ve been joined by a few converts along the way. However, I do understand how difficult it is to be the man driving against the traffic on an apparently one way street!
So with top-dressing already enjoying quite a bit of coverage on the site, I will leave it aside for the moment to concentrate on my other hint, which was the use of Bio-Liquid fertilisers.
But, first of all; what on earth are Bio Liquid Fertilisers?
A Bio (biological fertiliser) is one which contains carbohydrates in addition to the stated nutrients (N.P.K) often derived from minerals. The fertiliser can also contain other organic nutrients, amino acids etc.
Bio Fertilisers, of course, provide the plants with the basic building blocks we expect from fertilisers in the form of the essential soil nutrients, but the carbohydrate portion also nourishes the soil micro life (micro-organisms). This results in healthy plants and a healthy soil which allows us to produce better bowling surfaces consistently and predictably.
Research (Parent 1996) has shown that an active microbial flora means healthy plants, stable structures in the growth mass, reduced leaching of nutrients, and increased availability of a range of nutrients. Studies have also shown that micro-organisms – entirely naturally – produce antibiotics and vitamins which are actively absorbed by the plants.
As greenkeepers then, we should spend as much time thinking about the health of the soil as we do about achieving a fast, smooth surface as the two are inextricably linked. A simple and effective way of improving conditions for the micro life is to add carbohydrates, as a lack of available carbohydrates is the most limiting factor for the growth of soil micro-organisms (Parent 1996).
In addition to the above an active soil micro life is of major significance, as the micro-organisms contribute to the formation of stable soil aggregates via the discharge of polysaccharides. These are formed via adsorption of polysaccharides, which attach to clay particles. This is important on all greens, but the process is especially significant on sandy greens with low Cation Exchange Capacity (CEC), as it helps to reduce the leaching of nitrogen from the soil.
These polysaccharides form compounds with iron, aluminium, zinc and copper, and make them more available to the grass.
The micro-organisms also provide an invaluable service in breaking down organic material, which increases the breakdown of the thatch layer. This breaking down (mineralisation) releases nitrogen, sulphur, phosphorus, iron and other substances for the benefit of the plants.
With their content of carbohydrates, Bio fertilisers favour an active microbial flora. In comparison with traditional inorganic and organic fertilisers, this makes it easy for the greenkeeper to reduce the overall application rate of a range of nutrients, including nitrogen, allowing us to maintain our greens more economically and in a more environmentally aware manner.
We’ve seen that readily available carbohydrates are of major importance for the soil micro life, but the grasses also benefit directly from the application of carbohydrates – in particular, sucrose – which is available to them, as these can be absorbed to a minor extent directly by the leaves (Parent 1996).
A carbohydrate store in the plant is important throughout the entire growth season, as carbohydrates act as a direct source of energy in re-growth if/when the grass is exposed to injury, disease, stress and wear. The plant’s carbohydrate content is also an extremely important factor in the grasses ability to survive the winter (Beard 1973).

